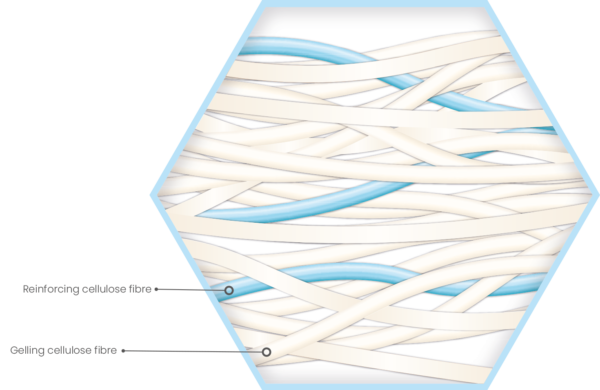
Modified Cellulose Dressings
Highly Absorbent with enhanced wet
strength
Sustainable
Fibres and fabrics directly derived from abundant and naturally occurring resources
Biocompatibility
Unique de-watering process which enhances the fibre strength, reduces fibre shed and provides increased biocompatibility.
Absorbency
Wet Strength
Optimised needle bonding process designed to balance the conflicting requirements of strength and absorbency
Product specifications
Our Carboxymethyl cellulose (CMC) and Cellulose Ethyl Sulfonate (CES) range can be supplied in roll form, slit reels or cut pieces, packaged and sterile. Various GSM available. CE marked and FDA approved. Antibacterial and antimicrobial modified cellulose options available.
For more details Click here to contact us.
- High absorbency
- High dressing strength and integrity (1)
- Enhanced comformability
- Reduced shrinkage (2)
- various GSM and cut sizes available on request
- Biodegradable material
- Moderately and highly exuding wounds
- Superficial or deep wounds such as:
- Pressure ulcers, leg and diabetic ulcers
- Surgical wounds
- Donor sites
- Exuding oncology wounds
All our products are manufactured in the UK, from class 8 clean room facilities located in Coventry. Our alginates, reinforced alginates, modified cellulose gelling fibre products are all manufactured from raw materials that are sustainable and using a manufacturing process which minimises waste as far as possible.
We can currently provide our products in several sizes from 5cm x 5cm cut pieces to 20cm x 30cm, although we can provide larger sizes on demand. We can provide our modified cellulose fabric in roll format, slit reels, cut pieces, ropes depending on requirements. These can be provided non-sterile or packed and sterile.
We keep some stock for the most commonly requested sizes for our x-ray detectable yarns, and therefore can deliver these on-demand. Most of our products are made to order, and whilst we often will work 12 weeks ahead with most of our customers to help manage a smooth continuity of supply which is particularly relevant with the current Covid-19 pandemic; We can often deliver in a few weeks and will try our best to meet your schedule. Packed and sterile products however will be between 8 to 12 weeks once the packaging has been fully approved.
SFM implements and complies with the ISO 13485 (EU, Canada, RoW), 21 CFR 820 (USA) and Ordinance 169 (Japan). We are experts in the overall planning and delivery of regulatory compliance throughout a product’s life cycle. We deliver products CE marked and or FDA cleared. Our Regulatory Affairs and Quality team manage our Integrated QMS.